JERSON FLORES C, ALEJANDRO ROSELL O.
Tipo:
Original Article
ABSTRACT (English):
Introduction: Astrocytoma are the most common tumors of the CNS and constitute 40 to 50% of all brain tumors. Survival depends on several clinical factors and treatment, and the actual survival of patients operated on astrocytoma grade II in our region is unknown, so the objective of the present study was to know the survival of patients operated on astrocytoma grade II as well as determine What is the impact of pre-surgical and post-surgical prognostic factors on the survival of patients with grade II astrocytoma that were operated at the Guillermo Almenara Hospital between 2003 and 2009.
Methods: A retrospective, observational and longitudinal study of 38 patients operated on astrocytoma grade II at the Guillermo Almenara Hospital between 2003 and 2009. The data was collected from medical records, operative reports and telephone interviews. The patients were classified according to the histological type of astrocytoma and the variables were analyzed: Group of prognostic risk, type of treatment and extension of the surgery. The SPSS 15.0 was used for the analysis.
Results: Of a total of 124 patients with astrocytoma, 30% (38/124) had a grade II astrocytoma, with an average survival of 56.7 months. According to the clinical prognosis group, survival of the low, medium and high-risk groups was 74.8, 46.0 and 31 months respectively. Regarding the type of treatment, the group with the highest survival was radiotherapy + surgery (66.2 months), followed by surgery alone (44.8 months) and the shortest survival time was radiotherapy alone (37.6 months). The longest survival was the total resection group (68.7 months), followed by the single biopsy (39.1 months) and the shortest group survived the partial resection (21.0 months).
Conclusions: Survival of patients operated on Astrocytoma grade II in our hospital is 56.7 months, with the factors of better prognosis being the "low risk" group and the total resection. Its classification into prognostic risk groups based on pre-surgical clinical data helps us predict survival before surgery.
Keywords: Astrocytoma, Prognosis, Brain Neoplasms, Survival, Risk factors (source: MeSH NLM)
ABSTRACT (Spanish):
Introducción: Los astrocitomas son los tumores más frecuentes del SNC y constituyen el 40 a 50% de todos los tumores cerebrales. La sobrevida depende de varios factores clínicos y de tratamiento, siendo desconocida la sobrevida real de los pacientes operados de astrocitoma grado II en nuestro medio por lo que el objetivo del presente estudio fue conocer la sobrevida de los pacientes operados de Astrocitoma grado II así como determinar cuál es el impacto de los factores pronósticos prequirúrgicos y postquirúrgicos en la sobrevida de los pacientes con astrocitomas de grados II que fueron operados en el Hospital Guillermo Almenara entre el 2003 y el 2009.
Métodos: Se realizó un estudio retrospectivo, observacional y longitudinal de 38 pacientes operados de astrocitoma grado II en el Hospital Guillermo Almenara entre 2003 y el 2009. Los datos fueron recolectados de historias clínicas, reportes operatorios y entrevistas telefónicas. Se clasificó a los pacientes según el tipo histológico de astrocitoma y se analizaron las variables: Grupo de riesgo pronóstico, tipo de tratamiento y extensión de la cirugía. Para el análisis se utilizó el SPSS 15.0
Resultados: De un total de 124 pacientes con astrocitoma, el 30% (38/124) tuvieron un astrocitoma grado II, siendo su sobrevida promedio de 56.7 meses. Según grupo de pronóstico clínico, la sobrevida de los grupos de bajo, mediano y alto riesgo fue 74.8, 46.0 y 31 meses respectivamente. Respecto al tipo de tratamiento el de mayor sobrevida el grupo de cirugía + radioterapia (66.2 meses), seguido por el de cirugía sola (44.8 meses) y el de menor sobrevida el de radioterapia sola (37.6 meses). Según extensión de la cirugía el de mayor sobrevida fue el grupo de resección total (68.7 meses), seguido por el de biopsia sola (39.1 meses) y el de menor sobrevida el grupo de resección parcial (21.0 meses).
Conclusiones: La sobrevida de los pacientes operados de astrocitomas grado II en nuestro hospital es de 56.7 meses, siendo los factores de mejor pronóstico el grupo de “bajo riesgo” y la resección total. Su clasificación en grupos de riesgo pronóstico en base a datos clínicos prequirúrgicos nos ayuda a predecir la sobrevida antes de una cirugía.
Palabras Clave: Astrocitomas, Pronóstico, Tumores cerebrales, Sobrevida, Factores de Riesgo (fuente: DeCS Bireme)
Astrocytomas are the m CNS and constitute 40 to 50% of all brain tumors, with malignant astrocytoma being the most predominant (70%). Surgery and radiotherapy are the main treatment options, but an optimal treatment regimen has not yet been established. The prognosis varies depending on several factors such as the histological grade, the clinical status and the type of treatment performed. Mortality is still high due to the fact that they are infiltrating tumors, so it is very difficult to do their total extirpation.
Numerous international studies have investigated the survival of patients undergoing surgery and radiotherapy and have determined the prognostic factors involved. Thus, in grade II astrocytoma it has been found that the factors with the best prognosis are age less than 40 years, absence of focal neurological deficit (epilepsy as the only symptom) and tumor less than 6 cm or that does not cross the midline.
However, despite being one of the most frequent brain tumors in the population whose surgical and complementary treatment is part of the daily neurosurgical practice in our country, there are no large studies that evaluate the real impact of surgery on survival and functional prognosis of patients with astrocytoma, taking into account prognostic factors such as histological grade, type of treatment and extension of surgery.
Knowing the survival of patients operated on in our region is important in order to offer a more realistic life expectancy, according to the patient's clinical status at the time of treatment. On the other hand, it is necessary to have a study that serves as a basis in the evaluation of the current treatment of astrocytoma, to be able to compare with subsequent studies and thus assess the progress in treatment based on the implementation of new surgical techniques or protocols. multidisciplinary management.
The objective of the present study was to determine the survival of patients with Astrocytoma Grade II who underwent surgery in our setting, as well as to determine the impact of pre-surgical (prognostic risk group) and postoperative factors (treatment and extension of surgery) in the survival of patients with grade II astrocytoma that were operated at the Guillermo Almenara Hospital between 2003 and 2009.
METHODS
A retrospective, analytical and longitudinal observational study was conducted in patients operated at the Guillermo Almenara Hospital between 2003 and 2009 and whose pathological diagnosis was grade II astrocytoma. The inclusion criteria were: Age over 18 years, anatomopathological diagnosis of grade II astrocytoma and supratentorial location. The exclusion criteria were: Age less than 18 years, diagnosis of astrocytoma not confirmed by pathological anatomy; and pathological diagnosis of oligodendrogliomas or oligoastrocytomas. The study was carried out between September and December 2012, and included all patients operated by grade II astrocytoma between January 2003 and December 2009.
Initially, 150 patients with an anatomopathological diagnosis of brain glioma were found. Of these, based on the inclusion and exclusion criteria, 38 patients with a diagnosis of low-grade II astrocytoma entered the study. The variables were grouped into 3 main groups: Dependent variables (survival and postoperative functional status), independent variables (histological type, type of treatment and extension of surgery); and comparison variables (age, location, preoperative functional status, focal neurological deficit, prognostic group and postoperative mortality).
Survival was defined as the time elapsed in months from the 1st surgery to the death of the patient due to causes related to the tumor. The postoperative functional status was determined according to the score (0 to 100) in the Karnosfky Scale at discharge. According to the score, 4 groups were established: Able to work (80-100), Able to be at home (50-70), Requires hospitalization (30-40), Requires permanent medical assistance (0-20). The histological grade according to the WHO classification in force during the years of the study and according to the Kernohan criteria, performed by a pathologist in a biopsy sample or operative piece. The grades are I, II, III, IV.
The type of treatment was defined as: Surgery (total, partial and biopsy total resection), Radiotherapy (after biopsy and administered by conventional fractionation), Surgery + Radiation therapy (surgery at the beginning and then adjuvant radiotherapy) and; Surgery + Radiotherapy + Chemotherapy (initial surgery, then adjuvant radiotherapy and chemotherapy, the latter from 2007)
The extension of the surgery was evaluated based on MRI or CT images before and after surgery (when available) or based on the surgical judgment of the surgeon as much as possible. Three groups were established: Broad Total Resection (resection without macroscopic residual tumor or more than 90% of initial tumor), Partial Resection (resection of less than 90% of initial tumor) and Biopsy, defined as resection of less than 10%, (open or with needle).
The focal neurological deficit was evaluated in 2 categories: Deficit: Clinical signs and symptoms, associated or not with epilepsy; and No deficit: Epilepsy as the only clinical manifestation.
The prognostic group is a composite variable that was defined by the combination of known clinical variables of prognostic significance in the survival of astrocytoma obtained from previous studies (such as EORTC or EOG). This depends on the histological type, and in the case of grade II astrocytoma, 3 groups were established: Low risk (age less than 40 and absence of focal neurological deficit), Medium risk (age less than 40 and presence of neurological deficit or older age / equal to 40 and absence of focal neurological deficit); and High Risk (age greater than or equal to 40 and presence of focal neurological deficit).
Operative mortality was defined as the number of patients who died between the day of surgery and day 30 after surgery.
Data collection was carried out based on clinical histories and operative reports. The telephone interview and direct observation were also used to determine the survival and the current status of the patient. The instrument used for data collection was a structured Ad hoc form. The analysis was carried out through the statistical program SPSS 15.0; using the Kaplan and Meier Method to determine the probability of survival over time and the Log Rank Test for the comparison of survival curves.
RESULTS
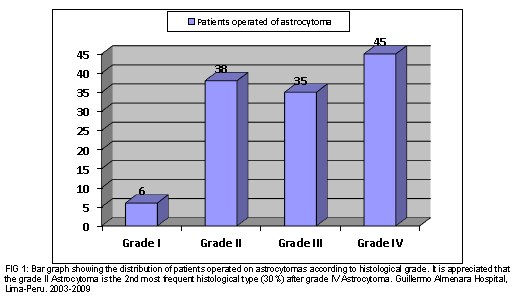
Between January 2003 and December 2009, 124 patients with cerebral astrocytoma were operated on, of which 30.6% (38/124) corresponded to a grade II astrocytoma. The rest were astrocytoma grade IV or glioblastoma 36.3% (45/124), astrocytoma grade III 28.2% (35/124) and only 4.8% (6/124) grade I astrocytoma or non-diffuse astrocytoma. (Fig 1)
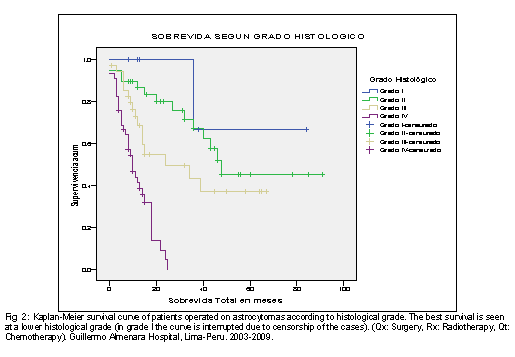
Regarding the global survival of the patients operated on astrocytoma according to the histological grade of the WHO, it was found that the survival of the patients with grade II astrocytoma was 56.7 months, being the survival in the grade I (68 months), 34.8 months in grade III and only 11.3 months in grade IV. This same trend is shown in Fig 2, which also shows its variation in time according to the survival curve of Kaplan and Meier.
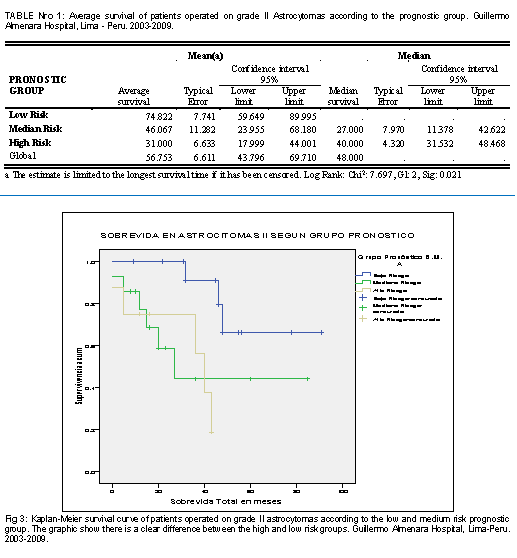
In relation to the survival of patients operated on astrocytoma grade II according to their clinical prognosis group (according to the previously established definition), it was found that survival in the low risk group was 74.8 months, followed by the medium risk group. with 46.1 months and the high risk with only 31.0 months. The degree of statistical significance was 0.021 (table No. 1). In Fig 3, the survival curves of the 3 clinical groups are shown, in which there is also a better survival of the low risk group with a clear difference with respect to the medium and high-risk group.
When evaluating the survival of patients with grade II astrocytoma according to type of treatment, it was found that the group with the best survival was Surgery + Radiotherapy (66.3 months), followed by Surgery alone (44.8), Radiotherapy (37.7 months) and the of Surgery + Radiotherapy + Chemotherapy (36.0 months) as observed in table No. 2. This same trend can be seen in the survival curves of the different groups (Fig 4). Although the group of Surgery + Radiotherapy + Chemotherapy has initially better Survival falls sharply after 36 months due to the end of the follow-up period.
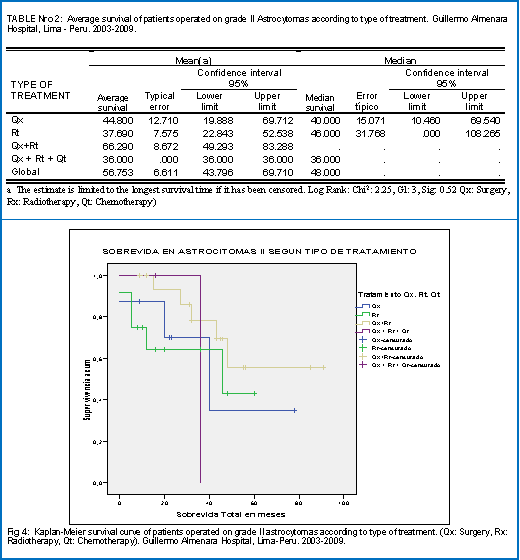
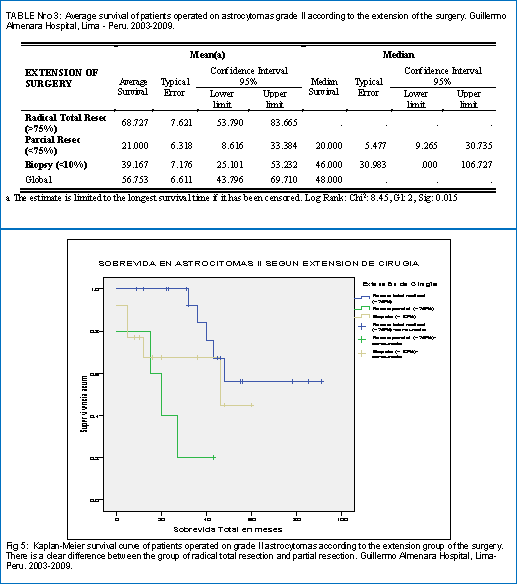
Table 3 shows the survival of patients with grade II astrocytoma according to the extent of the surgery. He found the best survival was obtained in the group of radical total resection (68.7 months), followed by the biopsy alone (39.2 months) and finally the partial resection (21.0 months). In fig 5, the survival curves corresponding to these 3 groups are appreciated, evidencing clearly a better survival of the radical total resection group with respect to the other 2 groups.
DISCUSSION
Astrocytomas constitute approximately 60% of glial tumors, and their frequency varies according to the histological grade: Grade I-10%, Grade II-16%, Grade III-20 to 25% and Grade IV-45 to 55% .1 The results of global survival vary in different international studies, but in general it is accepted that the average survival of patients with astrocytoma is: Grade I: 8 years (96 months), Grade II: 5 years (60 months), Grade III: 2.5 years (30 months), and Grade IV: 1 year (12 months) .2,3,4,5,6 This global survival varies depending on whether the patient has favorable or unfavorable prognostic factors.
In Peru, there are few publications about prevalence and survival in astrocytoma. Some of these national publications are those of Roca et al. Who published a series of 103 gliomas at the Guillermo Almenara Hospital in 1966, Landa et al. published another series of 100 brain gliomas between 1952-1962 and the most recent one published by Orrego Puelles in a series of 120 cases of astrocytoma of different degrees operated in the INEN between 1984-1994. In this last series, an average survival in astrocytoma of grades I-II of 43 months was found.7
In our study, we found that the overall survival of astrocytoma grade II was 56.8 months, which was higher than that reported in the national study of Orrego Puelles (56.8 vs 43 months). This result could be related to an overall improvement in the survival of patients with astrocytoma in recent decades, as demonstrated in the study by Sundeep Deorah et al.8 on trends in the prevalence and survival of brain cancer in the United States; to a better selection of patients or to an improvement in the treatment at the institutional level.9,10
Survival of patients with grade II astrocytoma depends on several prognostic factors, such as age, previous neurological condition, epilepsy as the only symptom, and the degree of surgical resection, which has been demonstrated in several clinical studies 12,13,14,15,18,19. Age and epilepsy as the only symptom have been 2 of the factors most strongly associated with a good prognosis.
In the present study 3 prognostic groups have been established, based on the union of several clinical factors in order to predict which subgroup has the best or worst survival after surgery. Thus, the low risk group includes patients with the best factors (age less than 40 and epilepsy as the only symptom), while the high risk group has the 2 negative factors (age over 40 and focal neurological deficit in addition to epilepsy), the medium risk being a combination of both, as previously described.
This is how it is demonstrated that the low risk group had a survival of 74.8 months higher than the average for this histological group (56.7 months) and higher than the survival of the medium and low risk groups, which was 46 and 31 months respectively, this difference being statistically significant (Sig 0.021).
According to the type of treatment received, the group of Surgery + Radiotherapy had the best survival (66 months) with respect to the groups of Surgery (44.8 months) and Radiation therapy alone (37.6 months) although the differences were not statistically significant (Sig 0.52). These results are in accordance with what is reported in the literature, because although some studies reported a favorable effect of radiotherapy, others such as the one carried out by the EORTC did not find a dose response relationship of radiotherapy in this histological group. 20 The shortest survival of the group of Surgery + Radiotherapy + Chemotherapy with respect to Surgery + Radiotherapy, probably due to a small number of patients as well as to a short follow-up period since in the survival curve there is an increased initial survival with respect to the other groups (which then falls sharply), which may be related to the beneficial effect of chemotherapy in a subgroup of grade II astrocytoma, as shown by some studies.15,21.
When evaluating the survival, according to the extension of the surgery, it is shown that the highest survival is obtained in the total resection group, whose survival (66 months) is greater than the average (56 months) and that of the partial resection groups ( 21 months) and only biopsy (39 months), the difference being statistically significant (Sig 0.015), which coincides with that reported in the literature of a better survival when the resection is totally radical, which was possible without considerably affecting the neurological function.
The present study has some limitations, these are related to the fact that we did not have at the beginning a standard method of histological classification of astrocytoma being informed only as low-grade astrocytoma, so it was necessary to review the sheets and a subsequent reclassification in degrees according to the WHO which may have affected the diagnostic accuracy.
Another limitation was the small size of the sample, so it is recommended to perform additional studies with larger samples that allow comparisons between subgroups of patients.
CONCLUSIONS
In patients with grade II astrocytoma, it is possible to predict the average survival before surgery based on clinical data grouped into groups of clinical prognoses. For this reason, it is recommended to classify patients with grade II astrocytoma according to clinical prognostic groups (low, medium and high risk) before surgery to have a better appreciation of the patient's life expectancy.
Likewise, the extension of the surgery has a significant impact on the survival, being the group with the largest total survival. The type of treatment also had a positive effect on survival, in favor of combined surgery and radiotherapy, although in this case the differences were not significant.
It is recommended to carry out further studies on survival of patients operated on astrocytoma that are compared with the current study, in order to assess progress in treatment by the implementation of new surgical techniques or protocols of multidisciplinary management. It is also necessary to have an updated database of patients with astrocytoma for a more accurate collection of information and better follow-up.
REFERENCES
-
Central Brain Tumor Registry of the United States [http://www.CBTRUS.org]
-
Rosalía Souto del Bosque, Salvador Aguilar Nieto, Adela Poitevin Chacón, Laura Suchil Bernal, Luis Dávila Maldonado, Miguel Ángel Celis López, Aída Mota García, Ernesto Gómez González. Astrocitomas cerebrales. Factores pronósticos en una serie de pacientes tratados en México. Revista de Oncología. Julio–Agosto 2001, Volume 3, Issue 4, pp 196-200.
-
Ohgaki, Hiroko PhD; Kleihues, Paul MD. Population-Based Studies on Incidence, Survival Rates, and Genetic Alterations in Astrocytic and Oligodendroglial Gliomas. Journal of Neuropathology and Experimental Neurology. Vol 64(6), Jun 2005, pp.
-
Lin CL, Lieu AS, Lee KS, Yang YH, Kuo TH, Hung MH, Loh JK, Yen CP, Chang CZ, Howng SL, Hwang SL.The conditional probabilities of survival in patients with anaplastic astrocytoma or glioblastoma multiforme. Surg Neurol. 2003 Nov;60(5):402-6; discussion 406.
-
Pignatti F, van den Bent M, Curran D, et al. Prognostic factors for survival in adult patients with cerebral low-grade glioma. J Clinical Oncology 2002; 20:2076-2084.
-
MLC van Veelen, C J J Avezaat, J M Kros,W van Putten, Ch Vecht. Supratentorial low grade astrocytoma: prognostic factors, dedifferentiation, and he issue of early versus late surgery. J Neurol Neurosurg Psychiatry 1998;64: 581–587
-
Orrego Puelles, José Enrique; Heinicke Yañez, Hugo Ricardo; Landa Cannon, Rodolfo. Manejo quirúrgico de los tumores intracraneales en el INEN: 1984-1993 / Surgical management of the intracranial tumors in the INEN: 1984-1993. Acta cancerol;24(3):27-30, sept. 1994.
-
Sundeep Deorah, M.A., Charles F. Lynch, M.D., Ph.D., Zita A. Sibenaller, Ph.D., Timothy C. Ryken, M.D. Trends in brain cancer incidence and survival in the United States: Surveillance, Epidemiology, and End Results Program, 1973 to 2001. Neurosurg Focus 20 (4):E1, 2006.
-
Efficacy and Toxicity of Postoperative Temozolomide Radiochemotherapy in Malignant Glioma. Martin Kocher, Sabine Kunze, Hans-Theodor Eich, Robert Semrau, Rolf-Peter Müller1. Strahlenther Onkol 2005 • No. 3
-
Takuma Nomiya, M.D., PH.D., Kenji Nemoto, M.D., PH.D.,Toshihiro Kumabe, M.D., PH.D., Yoshihiro Takai, M.D., PH.D., and Shogo Yamada, M.D., PH.D.. Prognostic significance of surgery and radiation therapy in cases of anaplastic astrocytoma: retrospective analysis of 170 cases. J Neurosurg 106:575–581, 2007.
-
Laws ER, Taylor WF, Clifton MB, et al. Neurosurgical management of low-grade astrocytoma of the cerebral hemispheres. J Neurosurg 1984;61: 665–73.
-
North CA, North RB, Epstein JA, et al. Low-grade cerebral astrocytomas. Survival and quality of survival after radiation therapy. Cancer 1990;66: 6–14.
-
Sofietti R, Chio A, Giordana MT, et al. Prognostic factors in well differentiated cerebral astrocytomas in the adult. Neurosurgery 1989;24: 686–92.
-
Medbery CA, Straus KL, Steinberg SM, et al. Low-grade astrocytomas: treatment results and prognostic variables. Int J Radiat Oncol Biol Phys 1988;15: 837–41.
-
Piepmeier JM. Observations on the current treatment of low-grade astrocytic tumors of the cerebral hemispheres. J Neurosurg 1987;67: 177–81.
-
Miralbell R, Balart J,Matias-Guiu X, et al. Radiotherapy for supratentorial low-grade gliomas: results and prognostic factors with special focus on tumour volume parameters. Radiother Oncol 1993;27: 112–6.
-
Janny P, Cure H, Mohr M, et al. Low grade supratentorial astrocytomas; management and prognostic factors. Cancer 1994;73: 1937–45.
-
Smith DF, Hutton JL, Sandeman D, et al. The prognosis of primary intracerebral tumours presenting with epilepsy: the outcome of medical and surgical management. J Neurol Neurosurg Psychiatry 1991;54: 915–20.
-
Recht LD, Lew R, Smith TW. Suspected low-grade glioma: is deferring treatment safe? Ann Neurol 1992;31: 431–6.
-
Karim A, Maat B, Hatlevoli R, et al. A randomized trial on dose response in radiation therapy of low-grade cerebral glioma: European organization for research and treatment of cancer (EORTC) study 22844. Int J Radiat Oncol Biol Phys 1996;36: 549–56.
-
J A. Pace, A. Vidiri, E. Galiè1, M. Carosi, S. Telera1, A. M. Cianciulli4, P. Canalini, D. Giannarelli5, B. Jandolo1 & C. M. Carapella1. Temozolomide chemotherapy for progressive low-grade glioma: clinical benefits and radiological response. Annals of Oncology 14: 1722–1726, 2003.
Disclosures
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Author Contributions
Conception and design: Flores, Rosell. Drafting the article: Flores. Critically revising the article: Flores. Reviewed submitted version of manuscript: Flores, Rosell. Approved the final version of the manuscript on behalf of all authors: Flores.
Correspondencia
Jerson M. Flores Castillo. Department of Neurosurgery. Guillermo Almenara National Hospital. 800 Grau. La Victoria. Lima 13, Perú. E-mail: jersonmit@yahoo.es