JOHN VARGAS U., ALFONSO BASURCO C., EDUARDO LAOS P., JESÚS CABREJOS B., RENZO ROMERO
Tipo:
Case Report
ABSTRACT (English):
Introduction: The morbidity of anterior cervical fusion surgery is low and one of the least frequent is an esophageal perforation, but this can bring severe complications such as sepsis, mediastinitis, or death. Optimal treatment is multidisciplinary, although it remains controversial; This consists of primary closure or closure by means of a muscle flap, accompanied by rest of the esophagus and a high protein diet.
Clinical Case: A 27-year-old female patient who, after a car accident, presented a cervical vertebromedullary trauma that required posterior cervical decompression surgery followed by stabilization via the anterior approach on 2 occasions. In the postoperative period, she presented erythema in the operative wound, subcutaneous emphysema, and cervical edema, for which she was re-admitted to the operating room being diagnosed with esophageal fistula. Treatment was performed by placing a muscle flap, antibiotics, esophageal bypass, and a high protein diet by nasogastric tube. The clinical and laboratory evolution was favorable, presenting closure of the esophageal fistula.
Conclusion: Esophageal fistula is a rare complication of anterior cervical surgery and this requires multidisciplinary management.
Keywords: Esophageal Fistula, Esophageal Perforation, Sepsis, Decompression, Spinal Fusion. (Source: MeSH NLM)
ABSTRACT (Spanish):
Introducción: La morbilidad de la cirugía de fusión cervical anterior es baja y una de las menos frecuentes es la perforación esofágica, pero esta puede traer complicaciones severas como sepsis, mediastinitis o la muerte. El tratamiento óptimo es multidisciplinario, aunque sigue siendo controversial; este consiste en el cierre primario o el cierre mediante un colgajo muscular, acompañado de reposo del esófago y dieta hiperproteica.
Caso Clínico: Paciente mujer de 27 años quien luego de un accidente automovilístico presentó un trauma vertebromedular cervical que requirió de cirugía de descompresión cervical posterior seguida de estabilización por vía anterior en 2 ocasiones. En el postoperatorio presentó eritema en herida operatoria, enfisema subcutáneo y edema cervical, por lo que reingresó a sala de operaciones siendo diagnosticada de fístula esofágica. El tratamiento se realizó mediante la colocación de un colgajo muscular, antibióticos, bypass esofágico y dieta hiperproteica por sonda nasogástrica. La evolución clínica y de laboratorio fue favorable presentando cierre de la fístula esofágica.
Conclusión: La fístula esofágica es una complicación poco frecuente de una cirugía cervical anterior y esta requiere un manejo multidisciplinario.
Palabras Clave: Fístula Esofágica, Perforación Esofágica, Sepsis, Descompresión, Fusión Vertebral. (Fuente: DeCS Bireme)
INTRODUCTION
In multiple studies, the morbidity rate for anterior cervical fusion ranges from 13.2 to 19.3%. The most frequent complications are dysphagia (1.7-9.5%), postoperative hematoma (0.4-5.6%), increase in the previous myelopathy (0.2-3.3%), symptomatic paresis of the recurrent laryngeal
nerve (0.9-3.1%), fluid fistula cerebrospinal (0.5-1.7%), soft tissue infection (0.1-1.6%), increased radiculopathy (1.3%), Horner syndrome (0.06-1.1%), respiratory failure (1.1%), esophageal perforation (0.3- 0.9%) and instrumentation failure (0.1-0.9%) 1, 7
A severe complication is perforation of the esophageal tissue, which can cause dysphagia, local soft tissue infection, deep tissue infection, and system failure, pseudarthrosis, osteomyelitis/discitis, sepsis, and in the worst cases infectious mediastinitis and death. 2
The optimal treatment for esophageal perforation is still controversial.3 Management is based on the time of diagnosis, size, the shape of the lesion, and general condition of the patient.4 Esophageal repair is done with support from colleagues in cardiothoracic surgery, otorhinolaryngology, and gastroenterology, 2, 4 being the most frequent the use of a muscle flap of the sternocleidomastoid (SCM), followed by the radial muscle of the forearm, pectoral muscle, omental flap, infrahyoid muscle, omohyoid muscle, latissimus dorsi muscle, and longus muscle of the neck. Second, there is the primary objection; and, finally, conservative treatment. 2
The clinical case of a patient with vertebromedullary trauma who suffered a postsurgical esophageal fistula is presented, which was successfully treated with the use of a muscle flap in addition to other support measures.
CLINICAL CASE
History and examination: 27-year-old female patient, a native of Trujillo and from Chiclayo, who, due to a traffic accident, suffered a cervical vertebromedullary trauma presenting paraplegia and brachial diparesis (strength in LL: 2/5 and UL: 1/5). The cervical tomography (CT) showed a C5 / C6 dislocation, for which she was operated on, performing a C5-C6-C7 decompressive laminectomy and a C6 corpectomy with iliac crest graft plus C5-C6-C7 anterior cervical fixation. She also underwent a tracheostomy due to respiratory failure due to in-hospital pneumonia, and she was later transferred to our Hospital.
The control cervical CT showed an inversion of the lordosis and invasion of the medullary canal due to graft migration, for which she was reoperated surgically, the removal of the C6 autologous graft, and a C5-C6 cervical fixation with a titanium graft. In postoperative 4, she presented erythema and signs of phlogosis in the operative wound, for which a surgical cleaning was performed where purulent discharge and gaseous content were found.
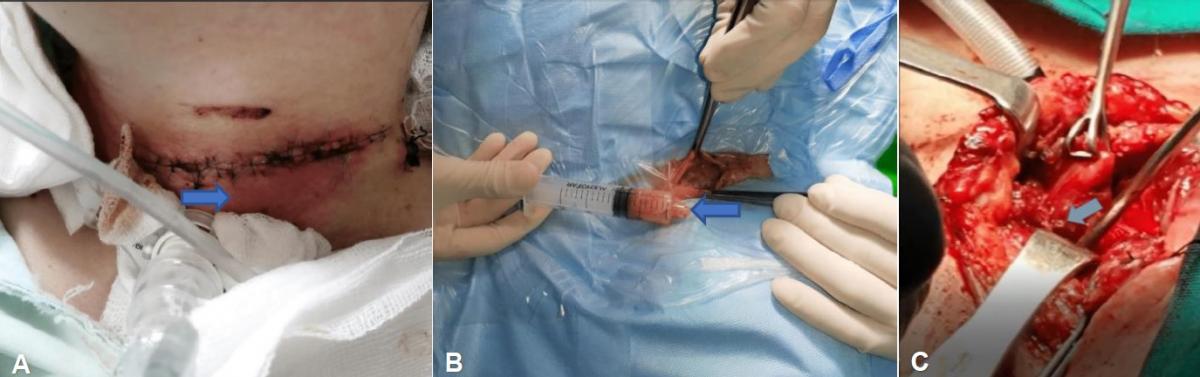
Treatment: In the following days, the patient presented with severe subcutaneous cervical and facial emphysema, being reoperated with the support of head and neck surgery. During surgery, an esophageal fistula was found due to perforation (<1 cm) in the dorsal aspect of the esophagus, for which it was decided to perform an SCM muscle flap in addition to a salivary bypass. (Fig 1)
|
Fig 1. (A) Image of the operative wound, showing local edema and signs of phlogosis (arrow). (B) Secretion obtained from the operative wound in the operating room, in his second intervention (arrow). (C) Intraoperative image of the punctate esophageal fistula on the dorsal aspect of the esophagus (arrow).
|
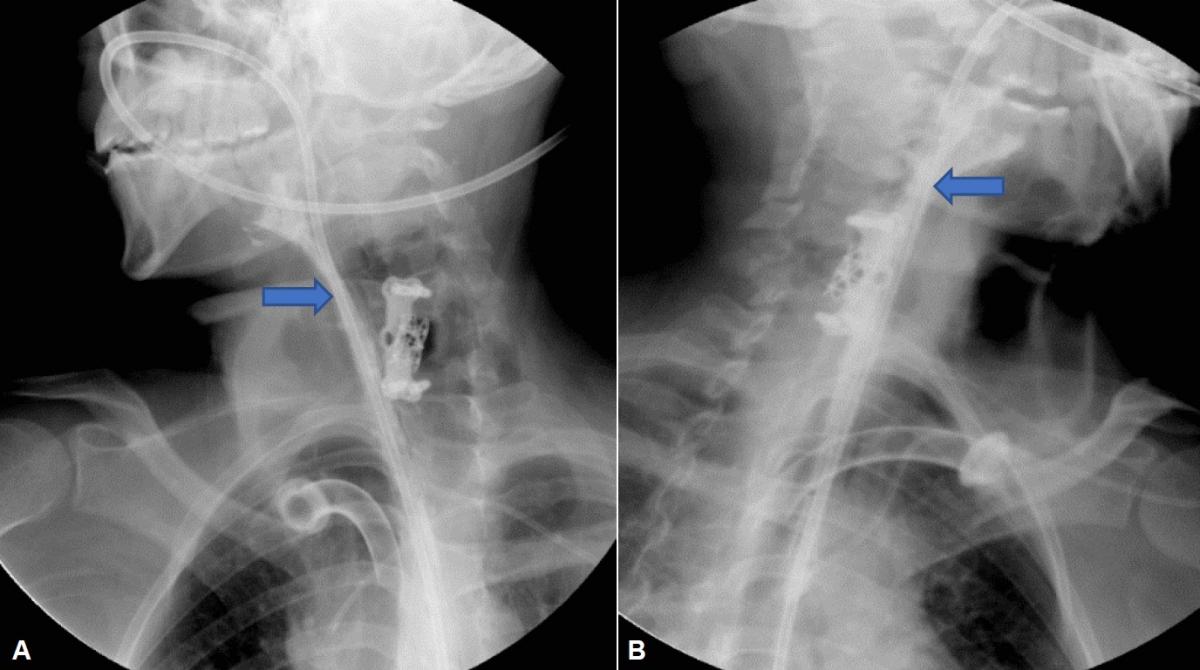
The culture of secretions was positive for Stenotrophomona maltophilia and Candida albicans, indicating treatment with trimethoprim/sulfamethoxazole and fluconazole, in addition to a high-protein and hypercaloric enteral diet by nasogastric tube, with this there was remission of fever and severe subcutaneous emphysema. A barium esophagogram showed the absence of an esophageal fistula. (Fig 2)
|
Fig 2. Barium esophagogram showing the adequate passage of contrast through the salivary bypass (blue arrow), from the proximal esophagus to the distal esophagus, without the presence of contrast leakage. (A) Right lateral view. (B) Left lateral view.
|
A cervical contrast-enhanced CT scan was performed which showed paratracheal abscesses. Broad-spectrum antibiotics (vancomycin and meropenem for 21 days) were indicated, thus presenting clinical improvement and reduction of abscesses in the control images. (Fig 3)
|
Fig 3. Cervical CT with contrast in coronal view showing bilateral paratracheal abscesses (thin arrows). (A) Tomography performed in postoperative 7 of the correction of the esophageal fistula. (B) Tomography performed in postoperative 14 of the correction of the esophageal fistula showing a decrease in the size of the abscesses described.
|
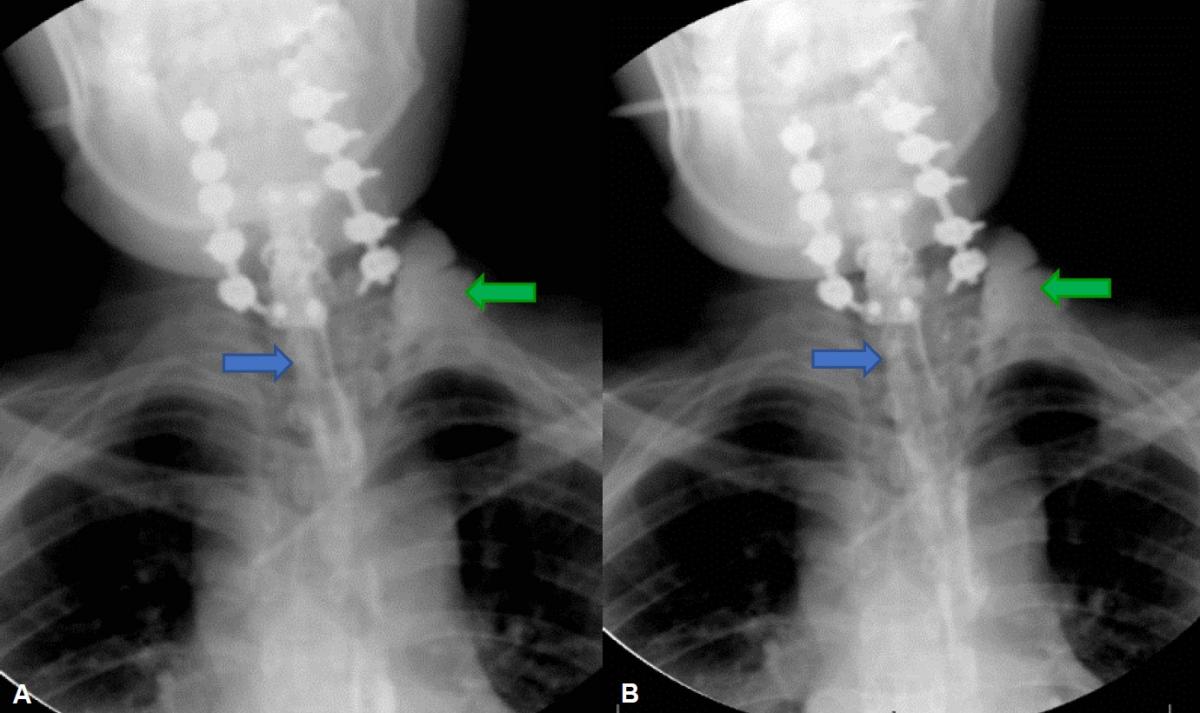
Then, a posterior fixation from C3 to C7 was performed with screws to lateral masses from C3 to C6 and a transpedicular screw to C7, in addition to removing the salivary bypass. A control esophagogram at 3 weeks showed slight contrast leakage towards the left cervical region, but without being a constant leak (Fig 4). The cervical collection remitted after evacuation and compression bandaging, in addition to receiving antibiotic treatment with ciprofloxacin and meropenem for an additional 10 days.
|
Fig 4. (A and B) Barium esophagogram showing contrast passage from the proximal to the distal esophagus without being able to show a fistulous trajectory (blue arrow), but if an image by addition in the left supraclavicular region (green arrow) can be evidenced which is suggestive of persistent esophageal fistula.
|
Clinical evolution: The patient evolved favorably after the closure of the fistula. She continued with high-protein and hypercaloric enteral nutrition for 2 more weeks. A new esophagogram did not show an esophageal fistula. The nasogastric tube was removed, and she began an oral diet.
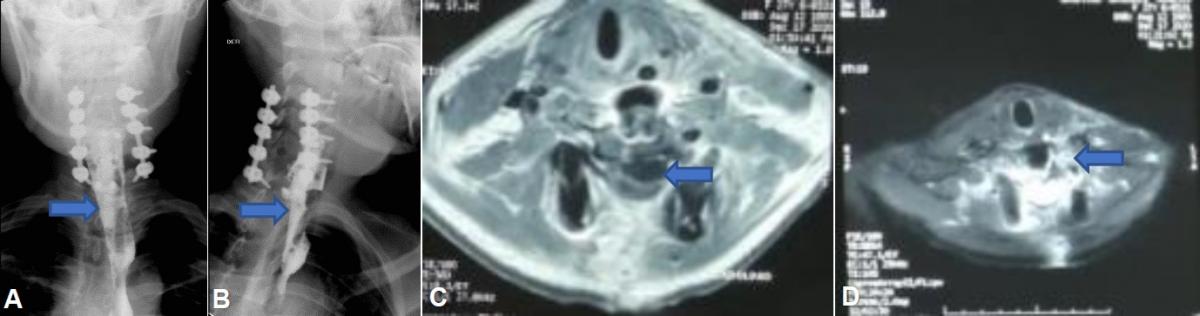
A cervical magnetic resonance imaging (MRI) with contrast showed old signs of the left anterior cervical inflammatory process and an uninfected post-traumatic cyst (Fig 5). The patient continued with good evolution, the muscular strength in UL improved slightly to a proximal predominance (Daniels 3/5), therefore, after 7 weeks of treatment, discharge and outpatient control were indicated.
|
Fig 5. (A) Barium esophagogram in anteroposterior view and in (B) lateral view, showing adequate contrast passage from the proximal esophagus to the distal esophagus (arrows), without evidence of fistula or addition images. (C) Cervical MRI in axial view and T2 sequence, where paratracheal abscesses are not evident, if a post-traumatic cyst is observed (arrow). (D) Cervical MRI with contrast in axial view and T1 sequence showing signs of old inflammation (arrow) in the left paracervical region without the presence of collections.
|
DISCUSSION
The anterior approach to the cervical spine, described in 1958, is safe and effective in treating both central and foraminal stenosis; For this, the natural planes of the cervical fascia are used, thus achieving an effective and safe exposure. 2, 7 In this, the common carotid artery and the internal jugular vein require mobilization and retraction so they can be damaged, although this is infrequent. A more frequent complication that may not be recognized is damage to the esophagus, which is in the superficial layer of the deep cervical fascia, dorsal to the trachea, and is lined by smooth muscle. 2, 4
Esophageal perforation during anterior cervical surgery is rare with an unknown prevalence, but with significant morbidity and mortality (3.92%).5 Its incidence varies between 0.02-1.52% and is higher even when the initial spinal injury is due to trauma (such as in our case), or there is a history of cervical surgery or irradiation. 2, 3 This data varies according to the author, Fountas et al report an incidence of 0.3%, while Zhong et al report 0.45%, and Epstein reports 0.3-0.9%. 1, 3 Esophageal perforation has a mortality of 0.1%, but the literature reports that it can be up to 16%, although most have a good prognosis. 1, 3
Halani et al in their case series found that the most frequent symptoms were dysphagia and odynophagia (63/117), followed by fever (24/117), cervical edema (23/117), and soft tissue fistula (18/117).2 Other symptoms that may occur are unexplained aspiration, hemoptysis, subcutaneous emphysema, or food residue in an operative wound, the latter being a pathognomonic finding.4 In our case, the symptoms that occurred were fever and progressive cervical edema, associated with subcutaneous emphysema which became severe (including the face), consistent with what was described in the medical literature.
Regarding the etiology, the most frequent cause of esophageal perforation is the failure of the system (41%), within it the migration of the plate, the migration of loose screws, plates, or screws are considered. The second most frequent cause is perforation due to chronic erosion by the system (31%). The third is the intraoperative injury (19%), which includes retraction and surgical instruments. And the fourth is the extrusion of the graft and subsequent penetration (7%). 2
Our case did not have a defined cause and it was considered to be due to a mixture of factors; on the one hand, the factor of a previous surgery that implies the presence of fibrosis and a greater risk of intraoperative injury; and, on the other hand, the presence of friable tissues in the surgical bed as a result of the over-aggregated infection.
The time of diagnosis is defined as the time between the initial cervical surgery and the diagnosis of esophageal perforation. Halani et al found a mean time to diagnosis of 716.6 days, with a median of 44.5 days, with a range from 0 days (intraoperative injury) to 18 years. An early perforation is one diagnosed in the first 30 days, as was our case, which was diagnosed in the first 96 hours. A late perforation is one that occurs after 30 days.2 Late perforations are mainly caused by migration or fracture of the fixation system (41%), followed by chronic erosion due to the mass effect of the fixation system (31%). Chronic pressure on the fixation system can cause tissue ischemia or the formation of a diverticulum, which weakens the posterior wall of the esophagus. 5
Multiple methods are used to diagnose the lesion, but the most widely used is the contrast swallowing study (esophagogram). CT, endoscopy, and MRI can also be used.2 Perrone et al mentioned that CT with contrast and cervical MRI is the Gold standard (92-100% sensitivity) as they allow the neck, mediastinum, spinal cord, and the fixing system. The contrasted esophagogram has a high degree of false negatives (25%) so it can be controversial.5 In our case, given the clinical worsening of the patient due to severe subcutaneous emphysema, the diagnosis was intraoperative by in situ vision of the small defect of the esophagus, although in our follow-up we also used CT, endoscopy, MRI, and esophagogram.
Regarding treatment, Ko et al, consider that the primary closure of the perforation and the prevention of infection is the Gold standard. If primary closure is impossible, then a muscle flap closure is performed with a double-layer suture.4 Some recommend conservative management of small perforations (less than 1cm), while surgery is preferred for injuries greater than 1cm. 5 Patients with a small perforation, with a well-contained fistula, without signs of sepsis, can undergo conservative treatment, which includes nothing by mouth for at least 1-week, broad-spectrum antibiotics, prokinetic drugs, and nasogastric tube feeding. 20-25% of patients develop abscess due to failure of this treatment, which increases mortality up to 18%. 4
Lee et al in their article published a therapeutic algorithm, where if the perforation is small (less than 1cm) and has a superficial vertebral defect (less than 3mm) and small (less than 3x3cm), primary closure or a flap of SCM muscle can be performed. On the other hand, if the perforation is small and the vertebral defect is deep (greater than 5mm), large (greater than 3x3cm), the dura is exposed, or osteomyelitis is present, then a primary closure with reconstruction using a flap of pectoral muscle should be performed. If the esophageal perforation is greater than 2cm or there is insufficient mucosa, we proceed to primary closure with reconstruction using a pectoral muscle flap. Lastly, if the defect is massive, a pedunculated flap of the longus colli muscle (long muscle of the neck) is performed associated with partial esophagectomy. 5
In addition to surgery, an aggressive diet should be given in terms of proteins (1.7-2g/kg/day) and calories, as well as normalizing thyroid function.5 In our case, what is mentioned in the literature was followed, and He proceeded to perform flap closure of the SCM muscle, rest of the esophagus, and a high protein and hypercaloric diet, achieving good clinical and imaging evolution. On the other hand, jejunostomy is recommended when it is necessary to keep the nasogastric tube for more than 2 weeks. If the cause is trauma due to the fixation system, the system should be removed, and a posterior cervical fixation proceeded. 4
In the event of a persistent esophageal fistula, its urgency (due to abscesses or mediastinitis) should be assessed. If it is not urgent, the thyroid profile and albumin levels should be evaluated, if they are altered, they should be corrected and the esophagogram repeated in 1-2 weeks. If it is urgent, or these values are normal, surgery should be reoperated to explore, review the flap, or enlarge with another flap.5 In our case, we continued with 2 more weeks of esophageal rest and a high protein diet, achieving closure of the esophageal fistula. Among the complications secondary to esophageal perforation, pneumonia, mediastinitis, osteomyelitis, sepsis, acute respiratory distress syndrome, and recurrent laryngeal nerve damage are mentioned,2 of which our patient, fortunately, did not suffer.
CONCLUSION
Esophageal fistula is a rare complication of anterior cervical surgery. It requires multidisciplinary management and should be treated as soon as possible to avoid greater morbidity and mortality and improve prognosis.
REFERENCES
-
Epstein N. A review of complication rates for anterior cervical diskectomy and fusion (ACDF). Surg Neurol Int. 2019; 10: 100.
-
Halani S, Baum G, Riley J, Pradilla G, Refai G, et al. Esophageal perforation after anterior cervical spine surgery: a systematic review of the literature. J Neurosurg Spine. 2016; 25 (3): 285-91.
-
Tasiou A, Giannis T, Brotis A, Siasios I, Georgiadis I, et al. Anterior cervical spine surgery-associated complications in a retrospective case-control study. J Spine Surg. 2017; 3(3): 444-459.
-
Ko SB, Park JB, Song KJ, Lee DH, Kim SW, et al. Esophageal perforation after anterior cervical spine surgery. Asian Spine J. 2019; 13(6): 976-983.
-
Lee TS, Appelbaum EN, Sheen D, Han R, Wie B. Esophageal perforation due to anterior cervical spine hardware placement: case series. Int J Otolaryngol. 2019; 7682654.
-
Yee TJ, Swong K, Park P. Complications of anterior cervical spine surgery: a systematic review of the literature. J Spine Surg. 2020; 6(1): 302-322.
-
Dantas FL, Dantas F, Mendes P, Sandes B, Fonseca Filho G. Primary repair of esophageal perforation following anterior cervical fusion. Cureus. 2020; 12(11): e11590.
-
Lee HC, Chen CH, Wu CY, Guo JH, Chen YS. Comparison of radiological outcomes and complications between single-level and multilevel anterior cervical discectomy and fusion (ACDF) by using a polyetheretherketone (PEEK) cage-plate fusion system. Medicine (Baltimore). 2019; 98(5): e14277.
-
Lord EL, Cohen JR, Buser Z, Meisel HJ, Brodke DS, Yoon ST, et al. Trends, costs, and complications of anterior cervical discectomy and fusion with and without bone morphogenetic protein in the United States medicare population. Global Spine J. 2017; 7(7): 603-608.
-
Saifi C, Fein AW, Cazzulino A, Lehman RA, Phillips FM, et al. Trends in resource utilization and rate of cervical disc arthroplasty and anterior cervical discectomy and fusion throughout the United States from 2006 to 2013. Spine J. 2018; 18(6): 1022-1029.
-
Chen CC, Huang YC, Lee ST, Chen JF, Wu CT, Tu PH. Long-term result of vocal cord paralysis anterior cervical discectomy. Eur Spine J. 2014; 23: 622-626.
-
Hofstetter CP, Kesavabhotla K, Boockvar JA. Zero-profile anchored spacer reduces the rate of dysphagia compared with ACDF with anterior plating. J Spinal Disord Tech. 2015; 28: e284-290.
-
Li Y, Zhu QS, Liu JC, Wu YT. Acute cervical epidural hematoma, screw pullout, and esophageal perforation after anterior cervical corpectomy surgery: report of a case. Int Surg. 2015; 100: 334-340.
-
Fehlings MG, Smith JS, Kopjar B, et al. Perioperative and delayed complications associated with the surgical treatment of cervical spondylotic myelopathy based on 302 patients from the AOSpine North America Cervical Spondylotic Myelopathy Study. J Neurosurg Spine. 2012; 16: 425-32.
-
Taylor BA, Vaccaro A, Albert TJ. Complications of anterior and posterior surgical approaches in the treatment of cervical degenerative disc disease. Semin Spine Surg. 1999; 11: 337-46.
-
Ardon H, Van Calenbergh F, Van Raemdonck D, et al. Oesophageal perforation after anterior cervical surgery: management in four patients. Acta Neurochir (Wien). 2009; 151: 297-302.
-
Anandaswamy TC, Pujari VS, Shivanna S, Manjunath A. Delayed pharyngoesophageal perforation following anterior cervical spine surgery: an incidental finding. J Anaesthesiol Clin Pharmacol. 2012; 28: 139-40.
-
Quadri SA, Capua J, Ramakrishnan, et al. A rare case of pharyngeal perforation and expectoration of an entire anterior cervical fixation construct. Journal of Neurosurgery: Spine. 2017; 26(5): 560-566.
-
Hershman SH, Kunkle WA, Kelly MP, et al. Esophageal perforation following anterior cervical spine surgery: case report and review of the literature. Global Spine Journal. 2017; 7(1): 28-36.
-
Noordhoek I, Koning MT, Jacobs WCH, et al. Incidence and clinical relevance of cage subsidence in anterior cervical discectomy and fusion: a systematic review. Acta Neurochir (Wien). 2018; 160: 873-80.
-
Shriver MF, Lewis DJ, Kshettry VR, et al. Pseudoarthrosis rates in anterior cervical discectomy and fusion: a meta-analysis. Spine J. 2015; 15: 2016-27.
-
Ghirelli M, Molinari G, Rosini M, et al. Pharyngo-esophageal perforations after anterior cervical spine surgery: management and outcomes. World Neurosurg. 2020; 139: 463-473.
-
Almre I, Asser A, Laisaar T. Pharyngoesophageal diverticulum perforation 18 years after anterior cervical fixation. Interact Cardiovasc Thorac Surg. 2014; 18: 240-241.
Disclosures
The authors report no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper.
Authors Contributions
Conception and design: All authors. Drafting the article: Vargas. Critically revising the article: Basurco, Vargas. Reviewed submitted version of manuscript: Vargas. Approved the final version of the manuscript on behalf of all authors: Vargas.
Correspondence
John Vargas Urbina. Department of Neurosurgery. Guillermo Almenara National Hospital. 800 Grau Avenue. La Victoria. Lima 13, Perú. E-mail: johnkilin27@hotmail.com